what factors at higher altitudes often contribute to greater incidence of frostbite
Sickle Prison cell Disease
KEY CONCEPTS
![]() Sickle jail cell disease (SCD) is an inherited disorder caused by a defect in the cistron for β-globin, a component of hemoglobin, and is called a qualitative hemoglobinopathy. Patients tin accept one defective cistron (sickle prison cell trait that is not a disease) or two lacking genes (SCD).
Sickle jail cell disease (SCD) is an inherited disorder caused by a defect in the cistron for β-globin, a component of hemoglobin, and is called a qualitative hemoglobinopathy. Patients tin accept one defective cistron (sickle prison cell trait that is not a disease) or two lacking genes (SCD).
![]() Although SCD usually occurs in persons of African ancestry, other ethnic groups can be affected. Multiple mutation variants result in different clinical manifestations.
Although SCD usually occurs in persons of African ancestry, other ethnic groups can be affected. Multiple mutation variants result in different clinical manifestations.
![]() SCD involves multiple organ systems. Usual clinical signs and symptoms include anemia, hurting, splenomegaly, and pulmonary symptoms. SCD can exist identified by routine neonatal screening programs. Early on diagnosis allows early comprehensive intendance.
SCD involves multiple organ systems. Usual clinical signs and symptoms include anemia, hurting, splenomegaly, and pulmonary symptoms. SCD can exist identified by routine neonatal screening programs. Early on diagnosis allows early comprehensive intendance.
![]() Patients with SCD are at risk for infection. Prophylaxis against pneumococcal infection reduces death during childhood in children with sickle cell anemia.
Patients with SCD are at risk for infection. Prophylaxis against pneumococcal infection reduces death during childhood in children with sickle cell anemia.
![]() Hydroxyurea decreases the incidence of painful episodes, but patients treated with hydroxyurea should be carefully monitored.
Hydroxyurea decreases the incidence of painful episodes, but patients treated with hydroxyurea should be carefully monitored.
![]() Neurologic complications caused by vasoocclusion can atomic number 82 to stroke. Screening with transcranial Doppler ultrasound to identify children at adventure accompanied past chronic transfusion therapy programs have been shown to be benign in decreasing the occurrence of overt stroke in children with SCD.
Neurologic complications caused by vasoocclusion can atomic number 82 to stroke. Screening with transcranial Doppler ultrasound to identify children at adventure accompanied past chronic transfusion therapy programs have been shown to be benign in decreasing the occurrence of overt stroke in children with SCD.
![]() Patients with fever greater than 38.five°C (101.3°F) should be evaluated, and advisable antibiotics administered immediately, including coverage for encapsulated organisms, particularly pneumococcal organisms.
Patients with fever greater than 38.five°C (101.3°F) should be evaluated, and advisable antibiotics administered immediately, including coverage for encapsulated organisms, particularly pneumococcal organisms.
![]() Pain episodes can often be managed at home. Hospitalized patients crave parenteral analgesics. Analgesic options include opioids, nonsteroidal antiinflammatory agents, and acetaminophen. The patient characteristics and the severity of the hurting should decide the selection of agent and regimen.
Pain episodes can often be managed at home. Hospitalized patients crave parenteral analgesics. Analgesic options include opioids, nonsteroidal antiinflammatory agents, and acetaminophen. The patient characteristics and the severity of the hurting should decide the selection of agent and regimen.
![]() Patients with SCD should be followed regularly for healthcare maintenance bug and monitored for changes in organ functions.
Patients with SCD should be followed regularly for healthcare maintenance bug and monitored for changes in organ functions.
![]() Sickle cell syndromes, which tin be divided into sickle cell trait (SCT) and sickle cell affliction (SCD), are a group of hereditary atmospheric condition characterized past the presence of sickle cell hemoglobin (HbS) in red blood cells (RBCs). SCT is the heterozygous inheritance of one normal β-globin factor producing HbA and i sickle gene producing HbS (HbAS) gene. Individuals with SCT are asymptomatic. SCD can exist of homozygous or compounded heterozygous inheritance. Homozygous sickle prison cell hemoglobin (hemoglobin S) (HbSS) has historically been referred to as sickle prison cell anemia (SCA). The heterozygous inheritance of HbS with another qualitative or quantitative β-globin mutation results in sickle cell hemoglobin C (HbSC), sickle cell β-thalassemia (HbSβ +-thal and HbSβ 0-thal), and some other rare phenotypes.one,2
Sickle cell syndromes, which tin be divided into sickle cell trait (SCT) and sickle cell affliction (SCD), are a group of hereditary atmospheric condition characterized past the presence of sickle cell hemoglobin (HbS) in red blood cells (RBCs). SCT is the heterozygous inheritance of one normal β-globin factor producing HbA and i sickle gene producing HbS (HbAS) gene. Individuals with SCT are asymptomatic. SCD can exist of homozygous or compounded heterozygous inheritance. Homozygous sickle prison cell hemoglobin (hemoglobin S) (HbSS) has historically been referred to as sickle prison cell anemia (SCA). The heterozygous inheritance of HbS with another qualitative or quantitative β-globin mutation results in sickle cell hemoglobin C (HbSC), sickle cell β-thalassemia (HbSβ +-thal and HbSβ 0-thal), and some other rare phenotypes.one,2
Over the years, progress has been made in agreement the relationship between clinical severity and genotype, every bit well every bit the natural history of common morbidities associated with SCD. Ongoing research involves investigation of pharmacotherapies used to treat SCD. Recent advances in the care of SCD patients have increased life expectancy.ane–seven
SCD is a chronic disease with significant brunt for family and guild. Frequent hospitalizations tin can interrupt schooling and result in employment difficulties.8–x Acute complications of the disease can be unpredictable, rapidly progressive, and life threatening. Afterward in life, chronic organ harm and cerebral or emotional impairments can develop.1,two Because of the complication and gravity of the illness, it is essential that comprehensive care is bachelor to all patients and that all providers involved have a proficient agreement of the disease and its management.1 ,2 ,8 ,11
EPIDEMIOLOGY
![]() SCD affects millions of people worldwide and is almost common in people with African heritage.ane,12 The well-nigh mutual SCD genotype is HbSS (~60% to 65%), followed past HbSC (~25% to 30%), HbSβ +-thal and HbSβ 0-thal (~5% to 10%). Other variants account for less than one% of patients.1,2 The illness is common among those with ancestors from sub-Saharan Africa, Republic of india, Saudi Arabia, and Mediterranean countries.2,13 In the United states, nearly 90,000 Americans have SCD with a prevalence of 1 in 2,500 newborns, 1 in 500 African Americans, and 1 in 36,000 Hispanic births.1,14,15 About 2 million Americans take SCT with a prevalence rate of 1 in 12 African Americans and one in 100 Hispanics.sixteen
SCD affects millions of people worldwide and is almost common in people with African heritage.ane,12 The well-nigh mutual SCD genotype is HbSS (~60% to 65%), followed past HbSC (~25% to 30%), HbSβ +-thal and HbSβ 0-thal (~5% to 10%). Other variants account for less than one% of patients.1,2 The illness is common among those with ancestors from sub-Saharan Africa, Republic of india, Saudi Arabia, and Mediterranean countries.2,13 In the United states, nearly 90,000 Americans have SCD with a prevalence of 1 in 2,500 newborns, 1 in 500 African Americans, and 1 in 36,000 Hispanic births.1,14,15 About 2 million Americans take SCT with a prevalence rate of 1 in 12 African Americans and one in 100 Hispanics.sixteen
About 275,000 babies are born with SCD every year and 85% occur in Africa.2,12 The prevalence of SCD in the region is determined past the frequencies of SCT. The distribution of SCT reflects the survival advantage in regions (tropical areas) where malaria is endemic as the factor mutation offers partial protection against serious malarial infection. RBCs carrying the abnormal sickle hemoglobin prevent the normal growth and development of Plasmodium falciparum within RBCs. Individuals with SCT are more than probable to survive the acute malarial affliction whereas individuals with SCD-HbSS often nowadays with more astringent disease. The incidence of the sickle factor in a population correlates with the historical incidence of malaria and SCT results in partial resistance to the illness.1,two,12,17
The prevalence of SCD is highest in sub-Saharan Africa. Other areas where the sickle mutation tin can be found include the Arabian Peninsula, the Indian subcontinent, and the Mediterranean region. In Africa, the variants are Senegal (Atlantic Westward Africa), Benin (Primal West Africa), Bantu (Cardinal African Republic), and Cameroon. Arab-Indian haplotype is seen in sure areas of Saudi arabia and India.2,13 Haplotypes identified through newborn screening programs in the United states of america showed that the Republic of benin haplotype was the nigh frequent (63%), followed by Bantu (14%), Senegal (9%), Cameroon (4%), and Saudi Arabian (ii%).13 Genetic analysis shows that the mutation found in Arabic patients is unlike from the mutation in those of African descent. Sickle factor mutation variants take been associated with different geographic locations and may be responsible for variations in clinical manifestations.3,5,13
ETIOLOGY
Normal hemoglobin (hemoglobin A [HbA]) is composed of 2 α chains and two β chains (α two β 2). The biochemical defect that leads to the development of HbS involves the substitution of valine for glutamic acid as the sixth amino acrid in the β-polypeptide concatenation. Another type of abnormal hemoglobin, hemoglobin C (HbC), is produced by the substitution of lysine for glutamic acrid as the 6th amino acid in the β-chain. Structurally, the α-bondage of HbS, HbA, and HbC are identical. Therefore, it is the chemic differences in the β-chain that business relationship for sickling and its related sequelae.1–three
SCA or HbSS is the about common class of SCD and occurs when an private inherits both maternal and paternal β-globin alleles that code for the HbS Figures 82-ane to 82-4 show the probability of inheritance with each pregnancy for the offspring of parents with HbA, SCT, and HbSS. If both parents are carriers, the offspring will have a 25% risk of inheriting SCD and a 50% risk of SCT (Fig. 82–1). β-Thalassemia is a quantitative hemoglobinopathy resulting from a genetic defect in β-globin production. β-Thalassemia can be co-inherited with HbS and may vary from no β-globin production (β 0) to some β-globin product (β +). Individuals with HbSS and HbSβ 0-thal have a more severe course than those with HbSC and HbSβ +-thal.2,13 Equally discussed earlier, several haplotypes characterize the sickle gene, resulting in different clinical and hematologic courses. Included amid these types are the three virtually commonly found in the United states: the Bantu haplotype, characterized by severe disease; the Senegal haplotype, characterized by balmy disease; and the Benin haplotype, characterized by a grade intermediate to that of the other two haplotypes. Although at that place are a number of other haplotypes seen around the world, the major types outside of the U.s. include Saudi Arabian and Republic of cameroon, both with milder courses of illness.2,three,5
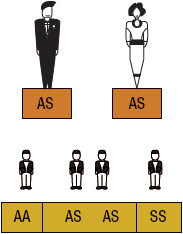
Effigy 82-1 Sickle prison cell gene inheritance scheme for both parents with sickle cell trait (SCT). Possibilities with each pregnancy: 25% normal (AA); l% SCT (AS); and 25% sickle cell anemia (SS). (A, normal hemoglobin; South, sickle cell hemoglobin)
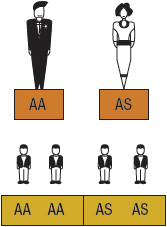
Figure 82-2 Sickle cell gene inheritance scheme for one parent with sickle jail cell trait (SCT) and 1 parent with no sickle cell cistron. Possibilities with each pregnancy: 50% normal (AA); l% SCT (AS). (A, normal hemoglobin; S, sickle cell hemoglobin)
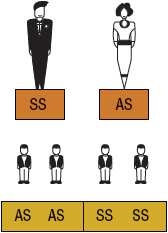
FIGURE 82-3 Sickle cell gene inheritance scheme for 1 parent with sickle prison cell trait (SCT) and one parent with sickle cell anemia (SCA). Possibilities with each pregnancy: fifty% SCA (SS); l% SCT (AS). (A, normal hemoglobin; South, sickle cell hemoglobin)
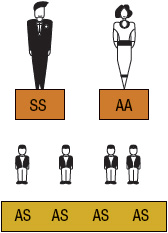
Figure 82-four Sickle cell inheritance scheme for one parent without sickle jail cell gene and 1 parent with sickle jail cell anemia (SCA). Possibilities with each pregnancy: 100% SCT (Equally). (A, normal hemoglobin; South, sickle cell hemoglobin)
PATHOPHYSIOLOGY
Normal adult RBCs incorporate predominantly HbA (96% to 98%). Other forms of hemoglobin are HbA2 (2% to 3%) and fetal hemoglobin (HbF; less than ane%). Normal RBCs are biconcave shape and able to deform to squeeze through capillaries.one–3,18 HbF is present predominantly in fetal RBCs and is a tetramer of 2 α globin chains and two γ globin chains (α two γ 2). Prior to nascence, HbF is the predominant hemoglobin type. Around 32 weeks gestation, a switch from the product of γ chains to β chains occurs and, consequently, an increase in HbA production is seen. Increased HbF production is seen nether severe erythroid stress, such as anemia, hematopoietic stem prison cell transplantation (HSCT), or chemotherapy or in the hereditary condition, hereditary persistence of fetal hemoglobin (HPFH) where a mutation in the β-globin factor cluster results in connected HbF production subsequently nascence. HPFH is a benign, asymptomatic condition.3,5,7,xix
In the pathogenesis of SCD, the following are primarily responsible for the diverse clinical manifestations: impaired circulation, destruction of RBCs, and stasis of claret flow. These changes event directly from two major disturbances involving RBCs: abnormal hemoglobin polymerization and membrane damage Fig. 82–5).
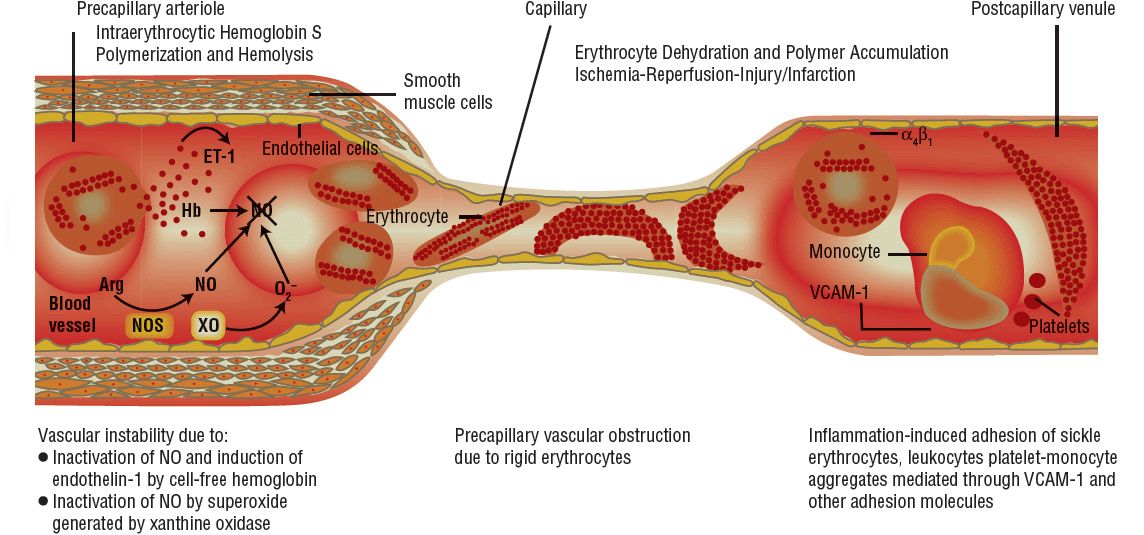
FIGURE 82-5 Pathophysiology of sickle cell disease. (Arg, arginine; ET-1, endothelin-i; Hb, hemoglobin; NO, nitric oxide; NOS, nitrous oxide synthase; VCAM-1, vascular prison cell adhesion molecule one; XO, xanthine oxidase.) (From Kato GJ, Gladwin MT. Sickle jail cell disease. In: Hall JB, Schmidt GA, Wood LDH, eds. Principles of Disquisitional Care, 3rd ed. New York: McGraw-Hill, 2005:1658.)
The solubilities of HbS and HbA are the same nether conditions of normal oxygenation. Because of increased hydrophobicity as a result of the valine-substituting glutamic acrid substitution, solubility of deoxygenated HbS is reduced. Saturation of deoxy-HbS leads to intermolecular binding and formation of sparse bundles of fibers, which initially are unstable. However, the increased binding of deoxy-HbS eventually results in cross-linked fibers and stable polymers. This process is influenced by mean corpuscular hemoglobin concentration (MCHC), temperature, intracellular pH, and the circulating amount of HbS. Polymerization allows deoxygenated hemoglobin molecules to exist equally a semisolid gel that protrudes into the cell membrane, leading to distortion of RBCs (sickle shaped) and loss of deformability. The presence of sickled RBCs increases blood viscosity and encourages sludging in the capillaries and postcapillary venules. Such obstructive events atomic number 82 to local tissue hypoxia, which tends to accentuate the pathologic process.one,2,six
When reoxygenated, polymers within the RBCs are lost, the RBCs eventually return to normal shape. This procedure contributes to the vasoocclusive manifestation in that HbS is able to squeeze into microvasculature when oxygenated, only becomes sickled when deoxygenated. The bicycle of sickling and unsickling results in harm to the prison cell membrane, loss of membrane flexibility, and rearrangement of surface phospholipids. Membrane harm also alters ion transport, resulting in potassium and water loss, which tin can lead to a dehydrated state that enhancing the formation of sickled forms. After continual repetitions of the procedure, the RBC membrane develops into rigid irreversibly sickled cell (ISC). Unlike the reversible sickled cells, which have normal morphology when oxygenated, ISCs are elongated cells and remain sickled when oxygenated. More rigid membranes of HbS-containing RBCs retard menses, particularly through the microcirculation. In addition, sickled RBCs tend to attach to vascular endothelial cells, which further increment polymerization and obstruction.one,2,6
Intermolecular bounden and polymer formation are reduced by HbF and to a lesser degree by HbAii. RBCs that comprise HbF sickle less readily than cells without. ISCs, not surprisingly, take a low HbF level. Increased levels of HbF, every bit in the instance of the Saudi Arabian genotype, effect in more benign forms of SCD. The amount of HbF and HbA2 in relation to HbS influences the clinical manifestations and accounts for some of the variability in severity amongst SCD genotypes.2,5
Intravascular destruction of sickle cells can occur at an accelerated rate. The stresses of circulation and repetitive sickle–unsickle cycles lead to prison cell fragmentation. Damage to the cell membrane promotes cell recognition by macrophages. Rigid ISCs are easily trapped, resulting in brusk circulatory survival and chronic hemolysis. The typical sickled cell survives for well-nigh 16 to 20 days, whereas the life bridge of a normal RBC is 120 days. Anemia triggers the release of young RBCs (reticuloctyes) from bone marrow prematurely. Surface adhesion proteins that maintain the reticulocytes inside the marrow adhere to the endothelium in postcapillary venules, further blocking the mature HbS-containing RBCs leading to complete apoplexy of microvessels.half-dozen,18
In improver to sickling, other factors are also responsible for the clinical manifestations associated with SCD. Hemolysis releases free hemoglobin resulting in generation of reactive oxygen species, and alternation of nitric oxide (NO) metabolism. Obstruction of blood period to the spleen by sickle cells tin can consequence in functional asplenia, divers as the loss of splenic part with an intact spleen. These patients tin can too have scarce opsonization. Dumb splenic role increases susceptibility to infection by encapsulated organisms, specially pneumococcal bacteria. Coagulation abnormalities in SCD can be the result of continuous activation of the hemostatic system or disorganization of the membrane layer.ii,6
CLINICAL PRESENTATION
![]() SCD is usually identified by routine neonatal screening programs in the United States. Since 2006, universal neonatal screening for SCD is performed in all fifty states. The sensitivity and specificity of screening methods (isoelectric focusing loftier-functioning liquid chromatography, or electrophoresis) arroyo 100%.20 For infants with a positive screening event, a second examination should be performed past 2 months of age to confirm the diagnosis. More than than 98% newborns in the United States are screened for SCD to identify the disease. Despite universal screening, some infants with SCD are not identified at nascency because of extreme prematurity, prior claret transfusion, or inability to contact family unit.19–21
SCD is usually identified by routine neonatal screening programs in the United States. Since 2006, universal neonatal screening for SCD is performed in all fifty states. The sensitivity and specificity of screening methods (isoelectric focusing loftier-functioning liquid chromatography, or electrophoresis) arroyo 100%.20 For infants with a positive screening event, a second examination should be performed past 2 months of age to confirm the diagnosis. More than than 98% newborns in the United States are screened for SCD to identify the disease. Despite universal screening, some infants with SCD are not identified at nascency because of extreme prematurity, prior claret transfusion, or inability to contact family unit.19–21
SCD involves multiple organ systems, and its clinical manifestations vary greatly betwixt genotypes Tabular array 82–1). Persons with SCT are usually asymptomatic and SCT is not considered a affliction. However, under certain extreme situations where hemoglobin oxygenation is contradistinct, RBC sickling tin occur. Individuals with SCT should be cautious when participating in exercise under extreme conditions, such equally high altitude or armed forces training. Sickling of RBCs in the renal medulla, an expanse with low oxygen tension, can outcome in the disability to concentrate urine. Individuals with such harm are at take a chance of aridity during periods in which the trunk ordinarily conserves h2o. Microscopic hematuria has been observed, and gross hematuria can occur after heavy exercise. Other reported complications associated with SCT are venous thromboembolism, renal medullary carcinoma, and chronic kidney disease.14,17,22 An increased incidence of urinary tract infection in women, peculiarly during pregnancy, was previously reported and specific screening guidelines should be followed.23
Tabular array 82-1 Clinical Features of Sickle Prison cell Trait and Common Types of Sickle Cell Disease
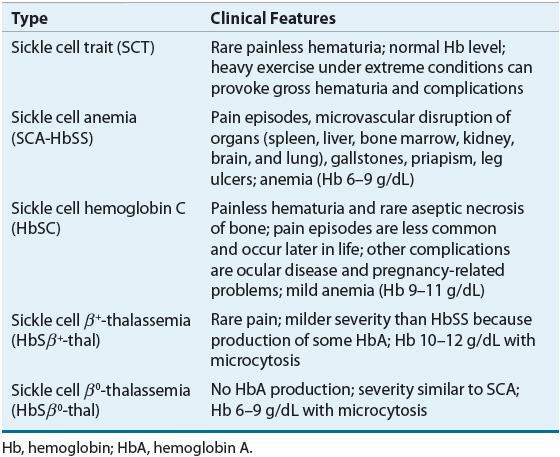
The cardinal features of SCD are hemolytic anemia and vasoocclusion. In individuals with HbSS, anemia commonly develops from four to 6 months after birth. The delay in presentation is due to the presence of HbF in the baby's RBCs. Even so, HbF production is gradually replaced by HbS, which typically leads the clinical manifestations of hemolysis and vasoocclusion, such as hurting and swelling of the easily and feet, ordinarily referred to every bit mitt-and-pes syndrome or dactylitis in infants.ane,two
The common clinical signs and symptoms associated with HbSS include chronic anemia and pallor; fever; arthralgia; scleral icterus; abdominal hurting; weakness; anorexia; fatigue; enlargement of the liver, spleen, and middle; and hematuria. Laboratory findings include the low hemoglobin level around half-dozen to 9 m/dL (3.72 to five.58 mmol/L), elevated reticulocytes of 10% to 20%, and elevated platelet and white claret cell (WBC) counts. Mean corpuscular volume (MCV) is normal. The peripheral blood smear demonstrates sickled red cell forms.1,fourteen,xviii
Individuals with HbSC illness nowadays with less astringent symptoms than that of HbSS and can exist characterized primarily by mild anemia (hemoglobin levels of 9 to eleven one thousand/dL [five.58 to vi.82 mmol/L] and reticulocytes of 3% to x%), infrequent episodes of hurting, persistence of splenomegaly into adult life, and excessive target cells in the peripheral blood smear. In individuals with heterozygous HbS-β-thalassemia syndrome, severity of illness depends on the thalassemia gene involved.ane,18
The Cooperative Written report of Sickle Cell Disease has previously reported that predictors for severe illness in children are dactylitis before 1 year of age, an average hemoglobin less than 7 g/dL (4.34 mmol/Fifty) in the second yr of life, and leukocytosis in the absence of infection. However, these variables could not be validated in a more than recent written report.24 Early acute chest syndrome (ACS) during the first 3 years of life is a predictor for recurrent episodes throughout childhood. Children with concomitant SCD and asthma have increased frequencies of ACS and pain episodes and increased bloodshed. Take chances factors for early death in adults with SCD include complications such every bit sickle cell pain, anemic events, ACS, renal failure, and pulmonary disease.1,25–27 With longer survival for SCD, chronic manifestations of the disease contribute to the increased prevalence of morbidity later in life.
COMPLICATIONS
Astute Complications
Fever and Infection
Functional asplenia and failure to brand antibodies against encapsulated organisms contribute to the loftier risk of overwhelming sepsis in individuals with SCD. Penicillin prophylaxis and vaccination have significantly reduced the overall risk of Streptococcus pneumonia bacteremia, simply an increased incidence of invasive pneumococcal infections with nonvaccine serotypes has been reported.28–30 Other encapsulated organisms are Haemophilus influenzae, Neisseria meningitidis, and Salmonella, and the latter has been known to cause osteomyelitis and pneumonia in SCD. Mycoplasma pneumoniae and Chlamydia pneumoniae should be considered in older children with infiltrates on chest radiograph. Viral infections (eastward.g., influenza and parvovirus B19) can result in severe morbidity.1,ii,18,28,29,31,32
All SCD patients with fever greater than 38.3°C (101°F) must be evaluated to decide the risk of infection or sepsis; workup should include physical examination, complete blood count with reticulocyte count, blood culture, breast radiograph, urinalysis, and urine culture. Lumbar puncture may be needed, especially in young and toxic-actualization children. A low threshold for empiric therapy compared to that in the general population is recommended.1,28,31,33
Children with SCD may experience a severe complication due to infection that results in impaired bone marrow production of RBCs. An aplastic crisis is characterized past a subtract in the reticulocyte count and the rapid development of astringent anemia (Table 82–two). The bone marrow becomes hypoplastic and is well-nigh frequently associated with a viral infection, particularly parvovirus B19.1,14,31
Tabular array 82-ii Acute Sickle Cell Complications

Neurologic
Neurologic abnormalities and cognitive deficits are well documented in patients with SCD. Vasoocclusive processes can atomic number 82 to cerebrovascular occlusion that manifests every bit signs and symptoms of overt stroke, such equally headache, paralysis, aphasia, visual disturbances, facial droop, and convulsions. The risk of stroke is highest for HbSS and lowest for HbSβ +-thal. The occurrence of cerebral infarct in HbSS is 11% by age xx years and 24% by historic period 45 years with a recurrence rate as high equally 70% in 3 years. The highest risk occurs during the first decades, in particular ages 2 to 5. The gamble is everyman before age two secondary to the protective event of HbF. Ischemic strokes occur in 54% of cerebrovascular accidents with the highest risk before age ten years and later 30 years of age; whereas hemorrhagic strokes are more common when patients are in their 20s and are associated with poor result.i,2,34
In addition to neurologic examination, evaluation of acute events include computed tomography (CT) scan and magnetic resonance imaging (MRI), magnetic resonance angiography for asymptomatic infarction, and transcranial Doppler (TCD) ultrasound to detect abnormal velocity and identify individuals at increased run a risk of stroke. In addition, electroencephalography (EEG) can be used if there is a history of seizure.i,2
Virtually 10% to thirty% of SCD who have HbSS with no prior history of stroke have been institute to have changes on MRI of the encephalon consistent with infarction or ischemia. These "silent infarcts" can be associated with increased risk of stroke, decreased neurocognitive functions, behavioral changes, and poor academic performances. Finally, lower intelligence, visual-motor impairments, and neuropsychological dysfunctions take been reported in patients not affected by acute or silent strokes and are associated with severity of anemia.1,34–37
Acute Breast Syndrome
ACS is the second most common cause of hospitalization and responsible for about 25% of expiry amidst individuals with SCD. ACS is defined equally a new pulmonary infiltrate associated with ane or more than of the post-obit: cough, dyspnea, tachypnea, chest pain, fever, wheezing, and new-onset hypoxia. As many equally half of individuals with SCD experience at least one episode of ACS. Risk factors for ACS and recurrence include young age (superlative incidence between age ii to 4 years), higher Hb, lower HbF, higher leukocytes, history of asthma or bronchial hyper-responsiveness, and smoke exposure. Genotype and haplotype as well influence the occurrence. Patients with HbSS and HbSβ 0-thal have higher incidence than those with HbSC and HbSβ +-thal. The prevalence is higher with African haplotypes than that of Saudi Arabia.1,25,38,39
The chief etiology for ACS is pulmonary vascular occlusion. The nigh common crusade of ACS is infectious and may be of bacterial, viral, or mycoplasma in origin. Infections, fatty emboli released from os marrow during pain crisis, or direct adhesion of RBC to the pulmonary vasculature leads to the inflammation and injury of the lung. Predictors for respiratory failure include all-encompassing lobar interest, platelet count less than 200,000 cells/mmiii (200 × 109/L), and history of cardiac or neurologic complications. In addition to physical examinations, evaluations should include breast radiographs. In severe cases, CT browse, perfusion scintigraphy, transthoracic echocardiography, and bronchoscopy should as well be considered. Pulmonary changes oft involve the lower lobes of the lungs and may crusade pleural effusions. Bilateral infiltrates or multiple lobe involvements may exist an indication of poor prognosis. The mortality rates for ACS are less than 1%, three%, and nine% for children upwards to nine years quondam, children 10 to xix years erstwhile, and adults, respectively. Pulmonary manifestations must exist recognized early and managed aggressively equally ACS can chop-chop progress to pulmonary failure and expiry.25,39,40
Priapism
Stasis and sickling of erythrocytes within the sinusoids of the corpora cavernosa is the primary mechanism of priapism, a sustained painful erection. In recent years, a meliorate understanding of pathophysiology of priapism has identified other mechanisms at the molecular level, such as NO and adenosine pathways. Stuttering priapism is repeated intermittent attacks upwards to several hours earlier remission; whereas ischemic priapism is a persistent painful erection greater than 4 hours and should exist considered as an emergency. 30 to 45% of males with SCD will nowadays with at least one episode of priapism during their lifetime and the first episodes often occur during childhood. Impotence has been reported after repeated episodes and is directly related to the elapsing prior to handling. ASPEN (association of sickle jail cell affliction, priapism, substitution transfusion, and neurologic events) syndrome has occurred one to xi days after fractional exchange transfusion in males who presented with priapism. This syndrome can range from headaches and seizures to obtundation requiring ventilation.41–43
Sickle Cell Hurting
Chronic hemolytic anemia in the SCD patient is periodically interrupted past astute episodes of hurting and vasoocclusion, particularly in childhood (Tabular array 82–2). Although fever, infections, dehydration, hypoxia, acidosis, and sudden temperature alterations can precipitate pain, multiple factors oftentimes contribute to its development.44
Sickle jail cell hurting may be caused by bone or muscle infarction due to vasoocclusion and may touch on a single site such as an arm, leg, or back or may involve multiple areas of the torso. Acute painful episode is the most common reason of hospitalization. Individuals with HbSS illness experience frequent episodes of pain than those HbSC disease or another variants.one,2,14 Biomarker for severity of pain during vasoocclusion episodes include C-reactive protein and lactate dehydrogenase (LDH).4,45 In addition, college pain score at presentation and older age are predictors for admission and longer elapsing of hospitalization.46 Dactylitis (hand-and-pes syndrome) is a sub-type of sickle cell pain, occurring in infancy and early childhood and is characterized by redness and swelling of the dorsal aspects of the hands, feet, fingers, and toes. The episodes are painful simply usually do non outcome in permanent harm.i
Splenic Sequestration
Splenic sequestration is the sudden massive enlargement of the spleen resulting from the sequestration of sickled RBCs in the splenic parenchyma (Table 82–two). Hematocrit and hemoglobin concentrations dramatically fall, with reticulocytosis and no prove of marrow failure or accelerated hemolysis. Trapping of the sickled RBCs by the spleen also leads to a decrease in circulating blood volume, which can outcome in hypotension and daze. The condition is most ofttimes seen in infants and children because their spleens are intact, and tin cause sudden death in immature children. Splenic enlargement may also be acutely painful due to rapid capsular expansion. Over time, repeated splenic infarctions lead to autosplenectomy and the spleen tin no longer become engorged; the incidence therefore declines every bit adolescence approaches.1,47
Chronic Complications
Pulmonary
Pulmonary hypertension as divers equally an elevated tricuspid regurgitation (TR Jet) velocity has been reported to be a run a risk cistron for death in adult patients with SCD.48 Diagnosis based on screening echocardiogram has been reported in children and adolescents, although its significance has not been established. Since recent studies have reported that screening echocardiogram alone has a high-false positive rate, pulmonary hypertension must be confirmed with right heart catheterization.48,49
The prevalence of asthma in children with SCD is similar to that of general population. However, asthma in individuals with SCD is associated with ACS and vasoocclusive pain episodes and increased mortality. Early on screening for asthma starting at historic period ane and pulmonary role test at least every v years starting at age 6 accept been recommended. Medications such as inhaled corticosteroids and β-agonists for asthma exacerbation should exist utilized for asthma control.1,27,48
Skeletal and Skin Diseases
Bone diseases are common in SCD and the vitamin D level has been suggested to exist the biomarker.4 Osteonecrosis, particularly of the femoral or humeral heads, causes permanent damage and disability.fourteen,50,51 Osteopenia associated with low bone formation has been reported in both males and females with SCD.52 Children with SCD also take an increased incidence of osteomyelitis; the organism near often responsible is Salmonella.31 In improver to necrosis of joints, chronic leg ulcers virtually commonly seen in the medial and lateral malleolus (ankles) tin go a difficult and painful problem for adult. Ulcers are oft seen after trauma or infection and are usually tedious to heal.53
Ocular Manifestations
Ocular problems seen in patients with SCD include transient monocular incomprehension, visual field defects from retinal hemorrhage, retinal detachment, vitreous hemorrhage, venous microaneurysms, and neovascularization. The incidence of proliferative retinopathy in SCD patients varies from 5% to ten%. Vasoocclusion in the eye can occur as early as 20 months of age, and clinically detectable retinal diseases normally occur during adolescence and early adulthood. Despite the less systemic manifestations, individuals with HbSC develop serious retinal complications more often and before than those with HbSS. Almanac test with retinal evaluation is recommended for patients with SCD to forestall blindness from retinopathy and other complications.54
Gastrointestinal Diseases
Cholelithiasis is a common complication of SCD resulting from chronic hemolysis and increased bilirubin production, and leading to biliary sludge and/or stone formation. The risk of gallstones increases with historic period: 14% by age 10 years and l% by age 22 years. Cholecystitis, exemplified past hurting in the right upper quadrant, can be dislocated with an acute sickle pain episode in the abdomen. Cirrhosis occurred in 18% of immature adults with SCD. Causes for development of chronic hepatic illness include repeated occlusion in the liver, fe overload, and hepatitis.1,14,55
Cardiac Diseases
Cardiovascular complications associated with anemia, including cardiac enlargement and various murmurs, tin occur in patients with SCD. Patients experience diverse degrees of exertional dyspnea, tachycardia, and palpitation because of the decreased oxygen-carrying chapters of the claret. Left ventricular diastolic dysfunction has been reported in 18% of adults with SCD and is associated with increased mortality, specially in patients with pulmonary hypertension. Left ventricular stiffness and left ventricular hypertrophy have been reported, and the progression is speculated to pb to diastolic dysfunction after in life. Acute myocardial infarction in adults with SCD may be underrecognized due to the high incidence of sickle cell acute chest pain.56,57
Renal
Sickling in the renal medulla, an area with relative hypoxia and low pH, can pb to sickle cell nephropathy. Renal dysfunction in SCD begins during infancy evidenced by glomerular hyperfiltration. Other manifestations include disability to concentrate urine, hematuria, tubular acidosis, papillary necrosis, glomerulonephritis, microalbuminuria, and proteinuria. Enuresis, as a consequence of increased urine production, is a common complaint. Chronic renal failure has been reported in xxx% of adults with SCD, and stop-phase renal disease is associated with high mortality.two,fourteen,58,59
Growth and Evolution
Delayed growth and sexual maturation are common in patients with SCD. Despite normal birth weight and length, growth retardation occurs betwixt 6 months and 4 years with height, weight, and bone mass alphabetize being affected. The poor growth cannot be explained by nutritional factors alone. Alternations in growth factors likewise as increased metabolic rate are factors contributing to the growth failure. Delay in puberty past 1 to ii years is mutual in adolescents with SCD and fertility bug tend to occur more oftentimes in female person SCD patients than in normal women.lx,61
Psychiatric
Depression and anxiety are more than common in children and adults with SCD than in the full general population and have a significant bear upon on quality of life.62,63 Depression is associated with hurting episodes in children with SCD. In add-on, the level of low has been reported to be associated with anxiety between mothers and children.64 DSM-Iv psychiatric diagnosis in adolescents with SCD include attention-deficit-hyperactivity, oppositional defiant, conduct, major depressive, and feet disorders.65
Pregnancy
Pregnancy introduces an increased risk for the mother with SCD and for the fetus. Some women feel increased pain episodes during pregnancy and the anemia of SCD can atomic number 82 to intrauterine growth retardation. Preterm labor and premature delivery are common in mothers with SCD, and the run a risk of spontaneous abortion is increased. The incidence of cesarean delivery and pregnancy-related complications are higher when compared to mothers who do not have SCD.14,66,67
Handling
Sickle Cell Disease
Desired Outcomes
The goal of treatment is to reduce hospitalizations, complications, and mortality. Handling for SCD involves the use of general measures to run across the unique demands for increased erythropoiesis. Additional interventions can be aimed at preventing or treating complications of the disease. When an acute complication occurs, the type and severity of the episode determine the appropriate therapeutic plan.
With availability of public health programs and comprehensive care, almost children survive through babyhood.12,26,68 The median survival rate is estimated to exist 42 years for males and 48 years for females for HbSS, and threescore years for males and 68 years for females for HbSC.12 With increased life expectancy, management of SCD should include assessment of health-related quality of intendance in both adults and children.
Patients with SCD crave lifelong multidisciplinary intendance. All patients with SCD should receive regularly scheduled comprehensive medical evaluations. Because of the complication of the disease, a multidisciplinary team is needed to provide high-quality medical care, teaching, counseling, and psychosocial support. Appropriate comprehensive care can have a positive impact on both longevity and quality of life. This care includes the utilize of evidence-based treatment combining general symptomatic supportive care, preventative medical therapies, and more specific affliction-modifying therapies aimed at altering hematologic capacity and function.
Routine Health Maintenance
Immunizations
Administration of routine immunizations is crucial preventive care in managing SCD. Impaired splenic role increases susceptibility to infection by encapsulated organisms, particularly Pneumococci. Prior to the routine utilise of penicillin prophylaxis and the development of pneumococcal vaccines, invasive pneumococcal disease was xx- to 100-fold more common in children with SCD than in healthy children. Reduced bloodshed has been associated with the introduction of pneumococcal vaccines.26,68
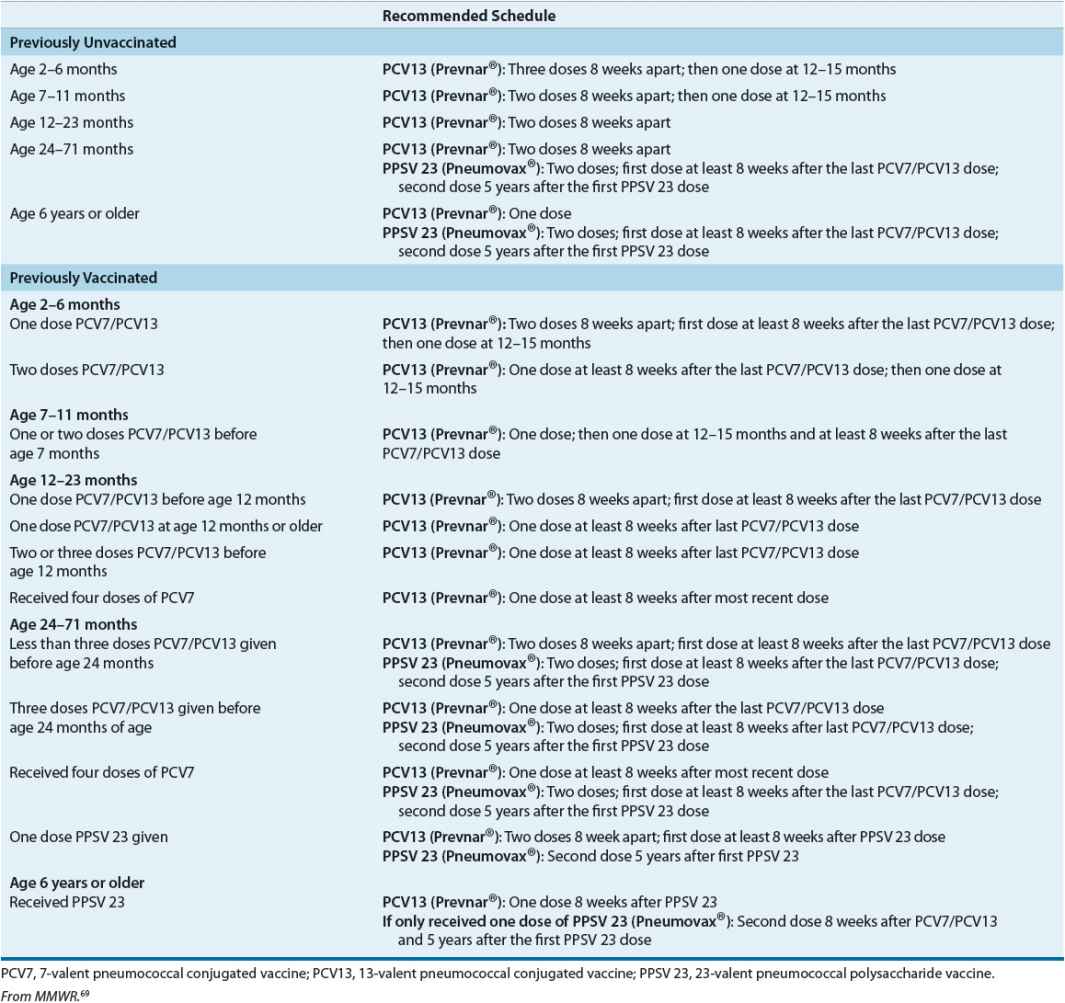
Two different pneumococcal vaccines are available. The 13-valent pneumococcal conjugate vaccine (PCV13; Prevnar®) induces good antibody responses in infants and children less than 2 years of age. Immunization with the PCV13 is recommended for all children, regardless of SCD status, younger than 24 months of age. Infants should receive the first dose after 6 weeks of age. 2 boosted doses should be given at two-month intervals, followed past a 4th dose at age 12 to 15 months. The 23-valent pneumococcal polysaccharide vaccine (PPSV 23; Pneumovax®23) is recommended for all children with functional or acquired asplenia but must exist given after 2 years of historic period because of poor antibody response. To cover different serotypes, PPSV 23 should be given starting at 2 years of age, and be administered ii months after the last dose of the 7-valent pneumococcal cohabit vaccine (PCV7)/PCV13. A booster dose of PPSV 23 is recommended 5 years after the first dose. The recommended immunization schedule and catch-up schedule for PCV13 and PPV 23 are presented in Tabular array 82–3. For children ages two to xviii years with SCD who did non receive PCV-13 during their routine immunization, a unmarried dose of PCV-xiii is recommended.69
Tabular array 82-3 Pneumococcal Immunizations for Children with Sickle Cell Illness
rodriguezbargeres89.blogspot.com
Source: https://basicmedicalkey.com/sickle-cell-disease-3/
0 Response to "what factors at higher altitudes often contribute to greater incidence of frostbite"
Enviar um comentário